We have learned how to defeat viruses quickly and effectively
COVID-19 belongs to the coronavirus family, with its reproduction mechanisms shared with the rest of RNA viruses. The on-going research concentrates on peculiarities of this particular virus’ genetic material and proteomics. NPO “International Medical Center SmartMed”, in collaboration with the leading Ukrainian Academy of Sciences institutes, has developed a unique multi-target antiviral therapy which utilizes a completely different approach, namely, targeting specific mechanisms affecting virus life cycle.
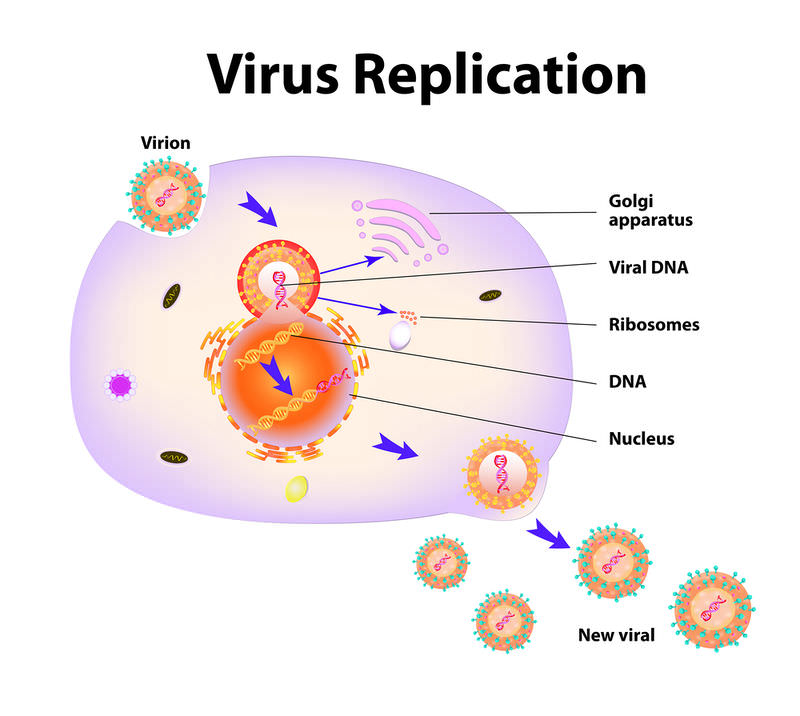
| Virus reproduction in a cell 1. Virus 2. Penetrates the cell and releases its own DNA or RNA 3. Virus DNA or RNA penetrates cell nucleus 4. Cell nucleus reproduces molecules of Virus DNA or RNA 5. New viral particles are assembled in the cell, destroying it and breaking out |
What you need to know about the virus:
- Every human body cell is a tiny factory that continuously produces proteins. Your genetic code (DNA) controls its every action. The DNA producing machine in the cell reads our own DNA and “makes sure” that cellular proteins are directed to where they are needed.
- A virus is an infectious agent, which has its own genetic code (DNA or RNA) enclosed in a protein shell (capsid), which protects genetic material during the virus “journey” from host to host. Besides, it assists in virus penetration into the host cell. There are surface proteins (in the case of coronaviruses, they do actually look like a crown) on the virus envelope coating, which serve the role of a key that “unlocks” entrance to the host cell.
- While viruses have their own genetic code, they have no mechanisms (equipment) which can copy viral DNA. They need a cell “factory” to clone themselves.
- How does the host cell takeover mechanism operate? In order to penetrate the cell, the virus uses surface proteins which bind to receptors on the host cell surface, thus enabling penetration of the cell membrane. Then, by releasing its own DNA or RNA into the infected cell, it essentially “captures”its protein factory like a “Trojan horse.”
The cell begins to produce and assemble viral genome and proteins. At this time, viral genetic material freely mixes with the host cell genes. This leads to active mutations of the virus and increases its survival abilities. While the virus is reassembled, it is “dressed up” in the new capsid. After being assembled, it leaves the cell and sets forth to “conquer new territories”. Viral particles separate and infect new cells, which then commence producing new viral clones.
Your cell no longer provides the intended physiological function; now it serves the virus by producing thousands of new viral particles which capture more and more cells into “slavery”. The contact of a viral particle with a cell membrane surface is the moment when a viral infection starts.
We have already mentioned virus mutation as one of the critical viral defence mechanisms. Such mutations of viral genetic material are exactly the reasons which prevent creation of effective vaccines or antiviral drugs targeting specific viral structures.
Antiviral therapy
- Silicon 3 is an antiviral drug which has a biophysics-based effect. Cerium dioxide particles of 2 nm in size are immobilized on the surface of finely dispersed silicon with a particle size of 150-200 nm. When colliding with viral particles, the drug particles of this particular size and charge trigger a reaction which damages the virus capsid and its surface proteins (here come lengthy and detailed physical explanations of long-range interaction forces, positive and negative particle charges, generation of atomic oxygen [1] and “potential energy wells”). In a nutshell, when the viral envelope/its surface proteins are damaged, the virus is not able to penetrate into a cell for further reproduction. At this point, its life cycle breaks.
Moreover, Silicon 3 has a powerful anti-inflammatory effect reducing proinflammatory interleukins and cytokines levels 10 times (notably, cytokine storm that occurs in the lungs is the primary cause of lung destruction in atypical coronavirus pneumonia).
In atypical pneumonia, alveoli are filled up with fluid and “cut out”, while fluid accumulation in the lungs leads to hypoxia. Silicon dioxide[2] increases body oxygenation by 2.5 times via facilitating oxygen delivery from alveoli to cell membrane. Silicon dioxide [3] promotes easier and more effective oxygen delivery in bodily tissues.
The drug is also a very powerful antioxidant. Numerous reactive oxygen species (ROS) overproduced in the course of inflammation are neutralized by Silicon 3. Due to the ability of silicon[4] dioxide to change valence and dioxidize[5] , 1 of its molecules interacts with 1 molecule of ROS, neutralizes it, deoxidizes and carries on (in contrast, 1 molecule of Vitamin C is also able to neutralize 1 molecule of ROS, but then it is also gone with no possible reactivation).
- Antivirus 7 is an antiviral drug equally effective against RNA and DNA viruses or, to be more precise, their reproduction mechanisms. Here we keep in mind that such viruses are still a protein shell enclosing genetic information.
The initial effect is that the drug contains tannin, which binds to viral surface proteins therefore blocking virus cell entry.
If the virus still manages to get into the cell, it is met by another surprise. Silicon particles with 2 nm size act as drug carriers, and exactly due to this size they easily “pull” the drug into the cell, which then inhibits RNA and DNA polymerases making virus “assembly” impossible.
If at this stage any virus manages to “survive”, the drug contains a specific enzyme that blocks intracellular protein responsible for “dressing” the virus in its capsid for future journey.
Moreover, in the case of chronic infection which affects immunocompetent cells, the drug triggers the infected cells apoptosis. Therefore, damaged non-effector cells are eliminated and replaced by their healthy counterparts, thus promoting immune system recovery.
To summarise, the drug blocks viral surface receptors so preventing virus entry into host cells. Next, it interferes with the processes of virus “construction” and “assembly”.
- Cytophil 4 acts as a powerful anti-inflammatory agent. It consists of 16 herbal extracts, including annual absinth sage, which is both a powerful anti-inflammatory agent and a cytostatic (used in oncology to stop cancer growth). So, adding it to the treatment scheme further suppresses the inflammation.
The proposed antiviral therapy must be started when the first symptoms of the disease manifest. In such a case, the fastest effect is observed. Administration of these drugs also prevents emergence of secondary bacterial infections.
As of April 29, 2020, we have used it to treat 89 patients with a confirmed “coronavirus infection” diagnosis. In 95% of cases the symptoms “went away” by day 5; starting from the 3rd day of the drugs administration significant health improvements were observed.
The results of our study are published in academic periodicals in Ukraine and abroad, and are also[6] patented:
V.I. Portnichenko, V.E. Dosenko, S.I. Pavlovich, А.М. Shish, Serebrovskaya Z.O.
Antioxidant, anti-inflammatory and metabolic effect Introduction of cerium dioxide nanoparticles in experimental pneumonia in rats.
V Scientific Conference for Nanoscale Systems: Structure, Properties, Technologies, NAS of Ukraine (Biofunctional nanomaterials, nanosystems in biology and medicine). December 1-2, 2016
Z. Serebrovska, V. Dosenko, A. Shish, S. Pavlovich, A. Dorovskych, V. Lysenko, V. Tertykh, J. Bolbuckh, V. Portnichenko Potential Application of Cerium Dioxide Nanoparticles for Acute Pneumonia Treatment. Wilderness Environ Med. 2017 March 2017 Volume 28, Issue 1, p1-62, e1-e6
http://www.wemjournal.org/article/S1080-6032%2817%2930014-5/fulltext
https://www.ncbi.nlm.nih.gov/pubmed/28531802Serebrovska Z, Swanson RJ, Portnichenko V, Shysh A, Pavlovich S, Tumanovska L, Dorovskych A, Lysenko V, Tertykh V, Bolbukh Y., Dosenko V. Anti – inflammatory and antioxidant effect of cerium dioxide nanoparticles immobilized on the surface of silica nanoparticles in rat experimental pneumonia. Biomed Pharmacother. 2017 May 19; 92: 69-77
Nanoparticles as antiviral agents against adenoviruses
Lysenko1, V. Lozovski1,2, M. Lokshyn1, Yu. V. Gomeniuk1,
A. Dorovskih3, N. Rusinchuk2*, Yu. Pankivska4, O. Povnitsa4,
S. Zagorodnya4, V. Tertykh5, Yu. Bolbukh5
https://imc.com.ua/nanoparticles-as-antiviral-agents-against-adenoviruses/
Нанофізика та антивірусна терапія В.Лисенко, В. Лозовський, М.Співак
https://imc.com.ua/nanofizika-ta-antivirusna-terapiya/
Near-field interaction explains features of antiviral action of non-functionalized nanoparticles
Valeri Z Lozovski, Volodymyr S Lysenko and Natalia M Rusinchuk
https://imc.com.ua/near-field-interaction-explains-features-of-antiviral-action-of-non-functionalized-nanoparticles/
Influence of the Virus-Nanoparticles System Illumination on the Virus Infectivity
O. Khylko, N. Rusinchuk, O. Shydlovska, M. Lokshyn, V. Lozovski, V. Lysenko, A. Marynin, A. Shcherbakov, M. Spivak, and N. Zholobak
https://imc.com.ua/influence-of-the-virus-nanoparticles-system-illumination-on-the-virus-infectivity/
Physical Point of View for Antiviral Effect Caused by the Interaction Between the Viruses and Nanoparticles
V. Lozovski, V. Lysenko, V. Piatnytsia, O. Scherbakov, N. Zholobak, and M. Spivak
https://imc.com.ua/physical-point-of-view-for-antiviral-effect-caused-by-the-interaction-between-the-viruses-and-nanoparticles/
Near-field Aspects of Antiviral Action of Complex Nanoparticles on Adenovirus
V. Lozovski and N. Rusinchuk, A. Dorovskih, V.Lysenko, M.Lokshyn, and Yu. Gomeniuk, Yu. Pankivska, O. Povnitsa, and S. Zagorodnya, V. Tertykh and Yu. Bolbukh
https://imc.com.ua/near-field-aspects-of-antiviral-action-of-complex-nanoparticles-on-adenovirus/
Nanoparticles in antivirus therapy
M. Lokshyn, V. Lozovski, V. Lysenko, V. Piatnytsia, M. Spivak and V. Sterligov
https://imc.com.ua/nanoparticles-in-antivirus-therapy/
Purification of Bioliquids from Viruses by Surface Plasmon-Polaritons
M. Lokshyn, V. Lozovski, V. Lysenko, Yu. Ushenin, N. Rusinchuk, O. Shydlovska, M. Spivak, and N. Zholobak
https://imc.com.ua/purification-of-bioliquids-from-viruses-by-surface-plasmon-polaritons/
Interaction between two nonpoint nanoparticles: Implementation to biology and medicine
V. Lozovski, V. Lysenko, V. Piatnytsia, M. Spivak
https://imc.com.ua/interaction-between-two-nonpoint-nanoparticles-implementation-to-biology-and-medicine/
Can nanoparticles be useful for antiviral therapy?
V. Lozovski, V. Lysenko, V. Pyatnitsia, M. Spivak
https://imc.com.ua/can-nanoparticles-be-useful-for-antiviral-therapy/
Interaction between viral particles and structured metal surface under surface plasmon propagation
V. Lozovski, V. Lysenko, M. Spivak, V. Sterligov
https://imc.com.ua/interaction-between-viral-particles-and-structured-metal-surface-under-surface-plasmon-propagation/
Z. Serebrovska, V. Dosenko, A. Shysh, S. Pavlovich, A. Dorovskych, V. Lysenko, V. Tertykh, J. Bolbuckh, V. Portnichenko Potential Application of Cerium Dioxide Nanoparticles for Acute Pneumonia Treatment. Wilderness Environ Med. 2017 March 2017 Volume 28, Issue 1, p1-62, e1-e6
Patent for invention No. 119489
Patent for invention No. 127738;
Patent for utility model No. 50803




